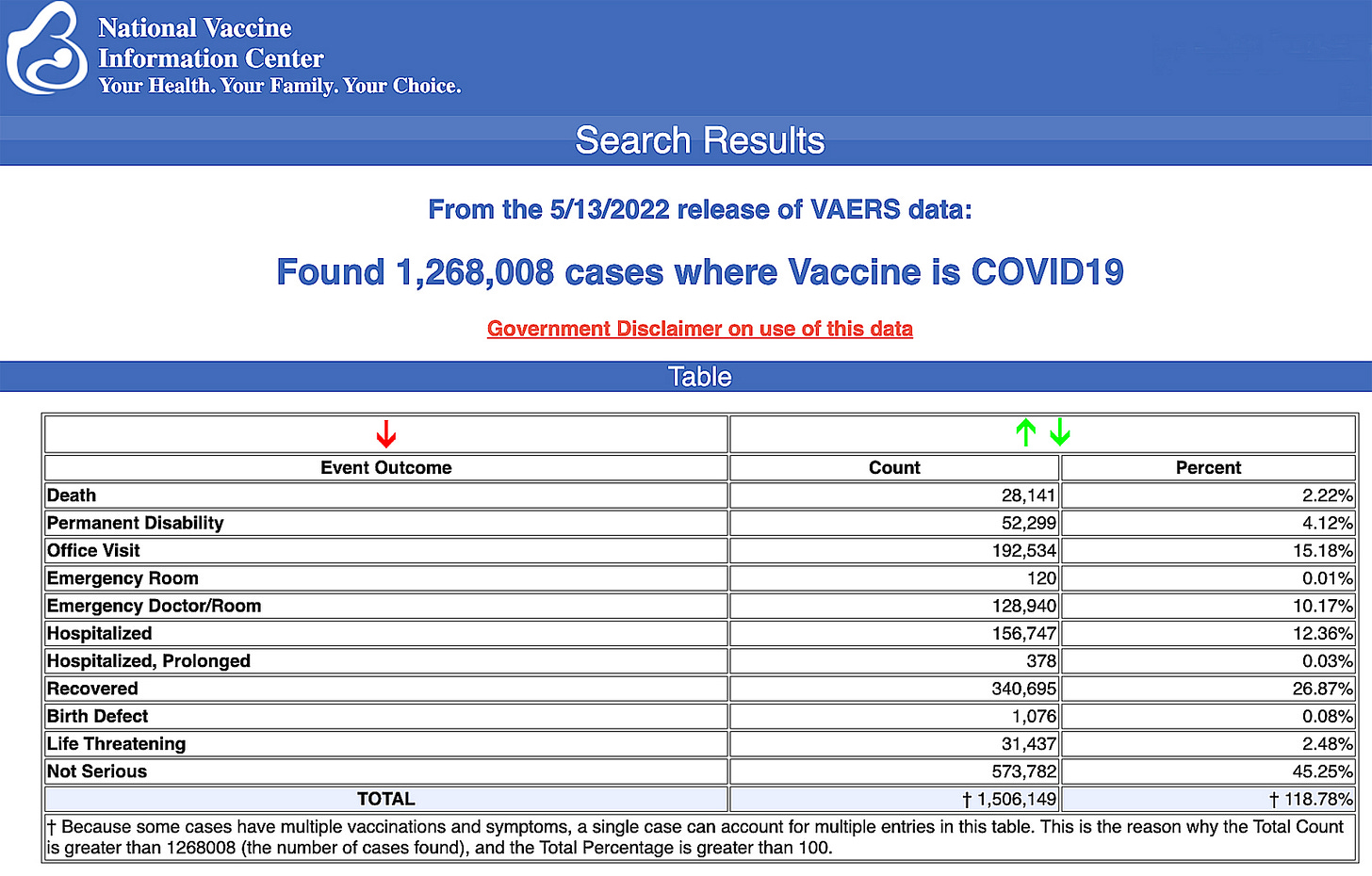
CDC's vaccine reporting VAERS has been shown to report only 1% of actual vaccine adverse events; now shows 1,268,008 adverse events, 28,141 deaths & 230,364 serious injuries Dec. 14, 2020-May 13, 2022
YET, despite this catastrophe of deaths and adverse events due to the COVID vaccine, CDC vaccine advisory panel follows FDA lead, endorses 3rd Pfizer shot for kids 5 to 11
“Adverse events from drugs and vaccines are common, but underreported. Although 25% of ambulatory patients experience an adverse drug event, less than 0.3% of all adverse drug events and 1-13% of serious events are reported to the Food and Drug Administration (FDA). Likewise, fewer than 1% of vaccine adverse events are reported.”
SOURCE:
What is the new updates:
Well, ‘U.S. VAERS data from Dec. 14, 2020, to May 13, 2022, for 5- to 11-year-olds show:
10,745 adverse events, including 279 rated as serious and 5 reported deaths.
22 reports of myocarditis and pericarditis (heart inflammation).The CDC uses a narrowed case definition of “myocarditis,” which excludes cases of cardiac arrest, ischemic strokes and deaths due to heart problems that occur before one has the chance to go to the emergency department.The Defender has noticed over previous weeks that reports of myocarditis and pericarditis have been removed by the CDC from the VAERS system in this age group. No explanation was provided.
43 reports of blood clotting disorders.
U.S. VAERS data from Dec. 14, 2020, to May 13, 2022, for 12- to 17-year-olds show:
31,572 adverse events, including 1,824 rated as serious and 44 reported deaths. VAERS reported 44 deaths in the 12- to 17-year-old age group last week.
64 reports of anaphylaxis among 12- to 17-year-olds where the reaction was life-threatening, required treatment or resulted in death — with 96% of cases attributed to Pfizer’s vaccine.
651 reports of myocarditis and pericarditis with 639 cases attributed to Pfizer’s vaccine.
168 reports of blood clotting disorders with all cases attributed to Pfizer.
U.S. VAERS data from Dec. 14, 2020, to May 13, 2022, for all age groups combined, show:
20% of deaths were related to cardiac disorders.
54% of those who died were male, 41% were female and the remaining death reports did not include the gender of the deceased.
The average age of death was 73.
As of May 13, 5,527 pregnant women reported adverse events related to COVID-19 vaccines, including 1,732 reports of miscarriage or premature birth.
Of the 3,622 cases of Bell’s Palsy reported, 51% were attributed to Pfizer vaccinations, 40% to Moderna and 8% to J&J.
880 reports of Guillain-Barré syndrome, with 42% of cases attributed to Pfizer, 30% to Moderna and 28% to J&J.
2,291 reports of anaphylaxis where the reaction was life-threatening, required treatment or resulted in death.
1,707 reports of myocardial infarction.
13,972 reports of blood-clotting disorders in the U.S. Of those, 6,266 reports were attributed to Pfizer, 4,989 reports to Moderna and 2,679 reports to J&J.
4,194 cases of myocarditis and pericarditis with 2,570 cases attributed to Pfizer’s, 1,427 cases to Moderna’s and 183 cases to J&J’s COVID-19 vaccines.
Children’s Health Defense (CHD) asks anyone who has experienced an adverse reaction, to any vaccine, to file a report following these three steps.
CDC vaccine advisory panel follows FDA lead, endorses 3rd Pfizer shot for kids 5 to 11
The CDC’s independent vaccine advisory panel on Thursday endorsed a third dose of the Pfizer-BioNTech COVID-19 vaccine for children ages 5 to 11.
CDC Director Dr. Rochelle Walensky signed off on the Advisory Committee on Immunization Practices (ACIP) recommendation, clearing the way for healthcare workers to begin administering the shots.
The ACIP recommendation came two days after the FDA granted Pfizer’s request for Emergency Use Authorization (EUA) of a single booster dose for this age group, saying the third shot should be administered at least five months after the initial two-dose primary series with the Pfizer-BioNTech vaccine.’

What the CDC is doing makes sense since they changed the definition of "vaccine" to suit their scams.
It's now up to the parents to surprise them in the court room.
Below is how you do it:
1. By 4 yogurts and ensure that the quantity is shown on the receipt
2. If anybody asks you for the proof of quackcination, show them the receipt and tell them that you got 4 shots.
3. If they refuse it, show them the definition of 'vaccine"
Your yogurts do match the CDC's definition of "vaccine" because
a) They do stimulate the body’s immune response against diseases like many other foods (Aloe Vera)
b) They're administered by mouth
c) You were quackcinated by introducing your yogurts into your body
---- CDC website
https://www.cdc.gov/vaccines/vac-gen/imz-basics.htm
Definition of Terms
Immunity: Protection from an infectious disease. If you are immune to a disease, you can be exposed to it without becoming infected.
Vaccine: A preparation that is used to stimulate the body’s immune response against diseases. Vaccines are usually administered through needle injections, but some can be administered by mouth or sprayed into the nose.
Vaccination: The act of introducing a vaccine into the body to produce protection from a specific disease.
Immunization: A process by which a person becomes protected against a disease through vaccination. This term is often used interchangeably with vaccination or inoculation.
we are coming for Fauchi, the FDA, CDC, the NIH, Big Pharma... absolute fact.. anyone found guilty of facilitating this Crime against humanity going to JAIL... Nuremberg